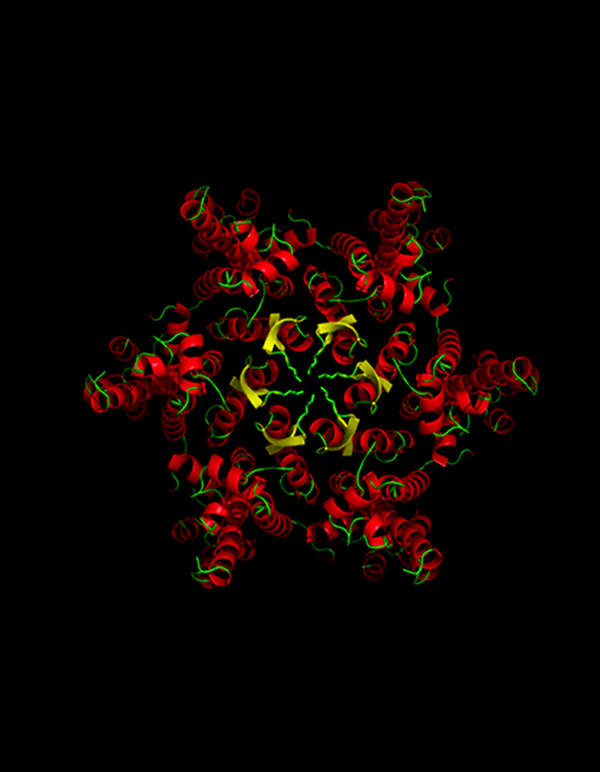
David Jacques is teasing apart the molecular interactions between viral and host proteins to figure out how viruses like HIV escape cellular defence networks.
Once viruses get into host cells, they need to run a gauntlet of molecular sensors and antiviral systems before they can replicate and spread to the next cell. Viruses have many ways of escaping detection: sometimes they outrun the defences, sometimes they hide, and sometimes they fight back.
David believes that one of the best ways to understand how viruses evade host defences—and discover new ways to stop viruses in their tracks—is to investigate the structures of the host and viral proteins while they are coupled together, using protein crystallography, or with cryo-electron microscopy.
Viruses exist because they have managed to find ways to subvert and control normal cellular functions. They achieve this through exquisitely specific molecular interactions with their hosts. David is interested in understanding the nature of these interactions. Understanding of host-virus interactions not only provides scope for the development of novel therapeutic antivirals, but often grants key insight into the molecular functions and defences of the host. Some examples of fundamental discoveries made through virus research include the discovery of oncogenes, restriction enzymes, and RNA sensing. David is particularly interested understanding the mechanisms by which viruses escape innate immune detection, and has a particular focus on HIV – a virus that specifically infects cells of the immune system.