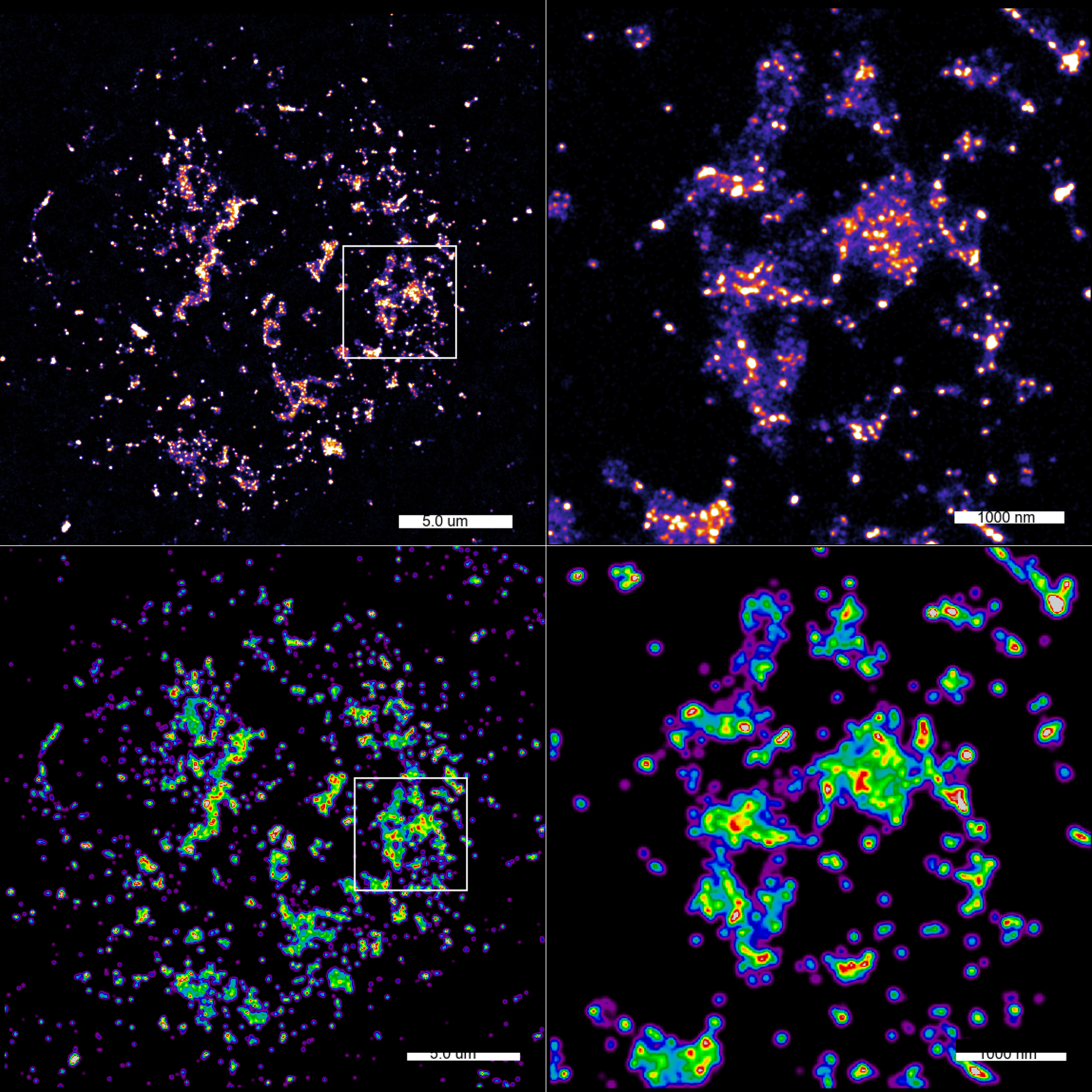
Jesse Goyette wants to understand exactly how T cells transmit signals to initiate an immune response. How does the T cell receptor work? What does a T cell need to be activated?
By making modifications to the signalling machinery, Jesse teases apart the function of individual components of the T cell receptor complex. T cells bearing modified receptors are imaged using super resolution microscopy, so we can see how molecules interact during a signalling event.
Jesse aims to identify which elements are essential for generating a robust T cell response, and figure out how each influence the properties of a T cell signal.
As T cells migrate through our bodies they continuously encounter antigen presenting cells and must be able to discriminate between foreign and self-antigens – initiating adaptive immune responses to the former, while remaining tolerant to the latter. The decision of a T cell to become activated or remain quiescent has important consequences and inappropriate reactions can lead to autoimmunity, whereas a lack of response leads to an inability to control infections or precancerous cells. A T cell’s decision-making process is encoded in the molecular language of proximal signalling reactions, which are integrated together to result in phenotypic responses such as cytokine production, differentiation, proliferation and release of cytotoxic granules.
Our research investigates the molecular mechanisms allowing signalling from multiple cell surface receptors to be integrated and result in different functional outcomes. To probe these mechanisms we use a combination of biochemical and biophysical techniques, such as recombinant protein production and surface plasmon resonance, together with advanced microscopy techniques such as total internal reflection fluorescence (TIRF) microscopy, direct stochastic optical reconstruction microscopy (dSTORM) and single particle tracking. Our approach also makes use of mathematical models, which are trained with information from our experimental results, to make predictions about outcomes of signalling processes that can in turn be used to guide future experimental work.
Understanding proximal signalling processes opens possibilities for manipulating the function of T cells and altering the outcome of inappropriate activation (in the case of autoimmune disease) or inappropriate tolerance (in the case of chronic viral infections or cancer).